Skeletal muscle tissue forms skeletal muscles. The best-known feature of skeletal muscle is its ability to contract and cause movement. Skeletal muscles act not only to produce movement but also to stop movement, such as resisting gravity to maintain posture. Small, constant adjustments of the skeletal muscles are needed to hold a body upright or balanced in any position. Muscles also prevent excess movement of the bones and joints, maintaining skeletal stability and preventing skeletal structure damage or deformation. Joints can become misaligned or dislocated entirely by pulling on the associated bones; muscles work to keep joints stable. Skeletal muscles are located throughout the body at the openings of internal tracts to control the movement of various substances. These muscles allow functions, such as swallowing, urination, and defecation, to be under voluntary control. Skeletal muscles also protect internal organs (particularly abdominal and pelvic organs) by acting as an external barrier or shield to external trauma and by supporting the weight of the organs.
Skeletal muscles contribute to the maintenance of homeostasis in the body by generating heat. Muscle contraction requires energy, and when ATP is broken down, heat is produced. This heat is very noticeable during exercise, when sustained muscle movement causes body temperature to rise, and in cases of extreme cold, when shivering produces random skeletal muscle contractions to generate heat.
Skeletal Muscle Structure:
Each skeletal muscle is an organ that consists of various integrated tissues. These tissues include the skeletal muscle fibers, blood vessels, nerve fibers, and connective tissue. Each skeletal muscle has three layers of connective tissue (called “mysia”) that enclose it and provide structure to the muscle as a whole, and also compartmentalize the muscle fibers within the muscle (Figure 1). Each muscle is wrapped in a sheath of dense, irregular connective tissue called the epimysium, which allows a muscle to contract and move powerfully while maintaining its structural integrity. The epimysium also separates muscle from other tissues and organs in the area, allowing the muscle to move independently.
Inside each skeletal muscle, muscle fibers are organized into individual bundles, each called a fascicle, by a middle layer of connective tissue called the perimysium. This fascicular organization is common in muscles of the limbs; it allows the nervous system to trigger a specific movement of a muscle by activating a subset of muscle fibers within a bundle, or fascicle of the muscle. Inside each fascicle, each muscle fiber is encased in a thin connective tissue layer of collagen and reticular fibers called the endomysium. The endomysium contains the extracellular fluid and nutrients to support the muscle fiber. These nutrients are supplied via blood to the muscle tissue.

Figure 1. The Three Connective Tissue Layers. Bundles of muscle fibers, called fascicles, are covered by the perimysium. Muscle fibers are covered by the endomysium.
In skeletal muscles that work with tendons to pull on bones, the collagen in the three tissue layers (the mysia) intertwines with the collagen of a tendon. At the other end of the tendon, it fuses with the periosteum coating the bone. The tension created by contraction of the muscle fibers is then transferred though the mysia, to the tendon, and then to the periosteum to pull on the bone for movement of the skeleton. In other places, the mysia may fuse with a broad, tendon-like sheet called an aponeurosis, or to fascia, the connective tissue between skin and bones. The broad sheet of connective tissue in the lower back that the latissimus dorsi muscles (the “lats”) fuse into is an example of an aponeurosis.
Every skeletal muscle is also richly supplied by blood vessels for nourishment, oxygen delivery, and waste removal. In addition, every muscle fiber in a skeletal muscle is supplied by the axon branch of a somatic motor neuron, which signals the fiber to contract. Unlike cardiac and smooth muscle, the only way to functionally contract a skeletal muscle is through signaling from the nervous system.
Skeletal Muscle Fiber (Cell) Structure
Because skeletal muscle cells are long and cylindrical, they are commonly referred to as muscle fibers. Skeletal muscle fibers can be quite large for human cells, with diameters up to 100 μm and lengths up to 30 cm (11.8 in) in the Sartorius of the upper leg. During early development, embryonic myoblasts, each with its own nucleus, fuse with up to hundreds of other myoblasts to form the multinucleated skeletal muscle fibers. Multiple nuclei mean multiple copies of genes, permitting the production of the large amounts of proteins and enzymes needed for muscle contraction.
The plasma membrane of a skeletal muscle fiber is called the sarcolemma. The sarcolemma is the site of action potential conduction, which triggers muscle contraction. Within each muscle fiber are myofibrils—long cylindrical structures that lie parallel to the muscle fiber. Myofibrils run the entire length of the muscle fiber, and because they are only approximately 1.2 µm in diameter, hundreds to thousands can be found inside one muscle fiber. They attach to the sarcolemma at their ends, so that as myofibrils shorten, the entire muscle cell contracts.

Figure 2. A skeletal muscle cell is surrounded by a plasma membrane called the sarcolemma with a cytoplasm called the sarcoplasm. A muscle fiber is composed of many fibrils, packaged into orderly units.
The cytoplasm of muscle fibers is referred to as sarcoplasm, and the specialized smooth endoplasmic reticulum, which stores, releases, and retrieves calcium ions (Ca++) is called the sarcoplasmic reticulum (SR) (Figure 2). 
Figure 2. Muscle Fiber. A skeletal muscle fiber is surrounded by a plasma membrane called the sarcolemma, which contains sarcoplasm, the cytoplasm of muscle cells. A muscle fiber is composed of many fibrils, which give the cell its striated appearance.
The Sarcomere
The functional unit of a skeletal muscle fiber is the sarcomere, a highly organized arrangement of the contractile myofilaments actin (thin filament) and myosin (thick filament), along with other support proteins. The striated appearance of skeletal muscle fibers is due to the arrangement of the myofilaments of actin and myosin in sequential order from one end of the muscle fiber to the other.
The sarcomere itself is bundled within the myofibril that runs the entire length of the muscle fiber and attaches to the sarcolemma at its end. As myofibrils contract, the entire muscle cell contracts. Because myofibrils are only approximately 1.2 μm in diameter, hundreds to thousands (each with thousands of sarcomeres) can be found inside one muscle fiber. Each sarcomere is approximately 2 μm in length with a three-dimensional cylinder-like arrangement and is bordered by structures called Z-discs (also called Z-lines, because pictures are two-dimensional), to which the actin myofilaments are anchored (Figure 3). Because the actin and its troponin-tropomyosin complex (projecting from the Z-discs toward the center of the sarcomere) form strands that are thinner than the myosin, it is called the thin filament of the sarcomere. Likewise, because the myosin strands and their multiple heads (projecting from the center of the sarcomere, toward but not all to way to, the Z-discs) have more mass and are thicker, they are called the thick filament of the sarcomere.
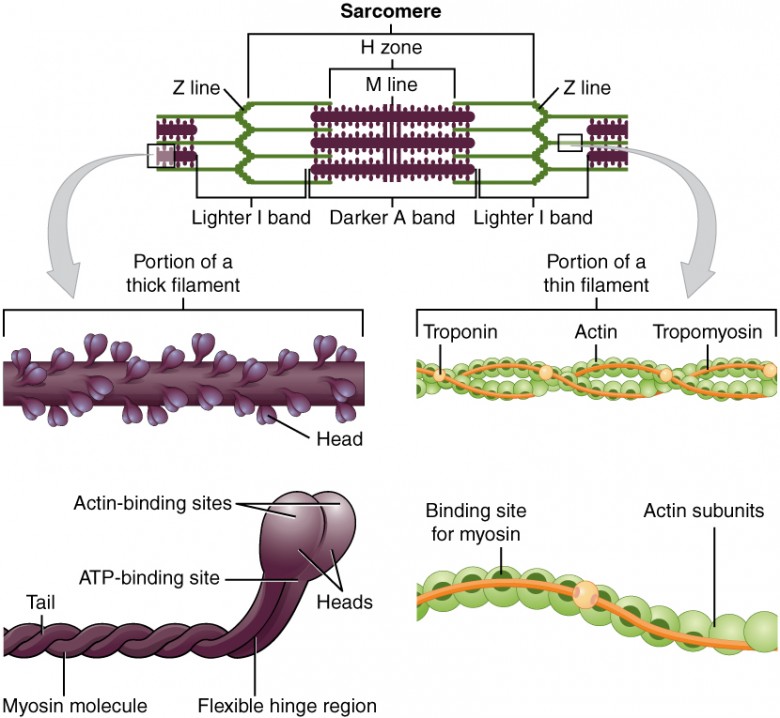
Figure 3. The Sarcomere. The sarcomere, the region from one Z-line to the next Z-line, is the functional unit of a skeletal muscle fiber.
Another specialization of the skeletal muscle is the site where a motor neuron’s terminal meets the muscle fiber—called the neuromuscular junction (NMJ). This is where the muscle fiber first responds to signaling by the motor neuron. Every skeletal muscle fiber in every skeletal muscle is innervated by a motor neuron at the NMJ. Excitation signals from the neuron are the only way to functionally activate the fiber to contract.
Signaling begins when a neuronal action potential travels along the axon of a motor neuron, and then along the individual branches to terminate at the NMJ. At the NMJ, the axon terminal releases a chemical messenger, or neurotransmitter, called acetylcholine (ACh). The ACh molecules diffuse across a minute space called the synaptic cleft and bind to ACh receptors located within the motor end-plate of the sarcolemma on the other side of the synapse. Once ACh binds, a channel in the ACh receptor opens and positively charged ions can pass through into the muscle fiber, causing it to depolarize, meaning that the membrane potential of the muscle fiber becomes less negative (closer to zero.) As the membrane depolarizes, another set of ion channels called voltage-gated sodium channels are triggered to open. Sodium ions enter the muscle fiber, and an action potential rapidly spreads (or “fires”) along the entire membrane to initiate the muscle to contract.
Things happen very quickly in the world of excitable membranes (just think about how quickly you can snap your fingers as soon as you decide to do it). Immediately following depolarization of the membrane, it repolarizes, re-establishing the negative membrane potential. Meanwhile, the ACh in the synaptic cleft is degraded by the enzyme acetylcholinesterase (AChE) so that the ACh cannot rebind to a receptor and reopen its channel, which would cause unwanted extended muscle excitation and contraction.
The Sliding Filament Model of Muscle Contraction
When signaled by a motor neuron, a skeletal muscle fiber contracts as the thin filaments are pulled and then slide past the thick filaments within the fiber’s sarcomeres. This process is known as the sliding filament model of muscle contraction (Figure 6).
The sequence of events involved in a muscle contraction:
The sequence of events that result in the contraction of an individual muscle fiber begins with a signal—the neurotransmitter, ACh—from the motor neuron innervating that fiber. The local membrane of the fiber will depolarize as positively charged sodium ions (Na+) enter, triggering an action potential that spreads to the rest of the membrane will depolarize, including the T-tubules. This triggers the release of calcium ions (Ca++) from storage in the sarcoplasmic reticulum (SR). The Ca++ then initiates contraction, which is sustained by ATP (Figure 4). As long as Ca++ ions remain in the sarcoplasm to bind to troponin, which keeps the actin-binding sites “unshielded,” and as long as ATP is available to drive the cross-bridge cycling and the pulling of actin strands by myosin, the muscle fiber will continue to shorten to an anatomical limit.
Muscle contraction usually stops when signaling from the motor neuron ends, which repolarizes the sarcolemma and T-tubules, and closes the voltage-gated calcium channels in the SR. Ca++ ions are then pumped back into the SR, which causes the tropomyosin to reshield (or re-cover) the binding sites on the actin strands. A muscle also can stop contracting when it runs out of ATP and becomes fatigued (Figure 5)

Figure 4. Contraction of a Muscle Fiber.

Figure 5. Relaxation of a Muscle Fiber.
The region where thick and thin filaments overlap has a dense appearance, as there is little space between the filaments. This zone where thin and thick filaments overlap is very important to muscle contraction, as it is the site where filament movement starts. Thin filaments, anchored at their ends by the Z-discs, do not extend completely into the central region that only contains thick filaments, anchored at their bases at a spot called the M-line. A myofibril is composed of many sarcomeres running along its length; thus, myofibrils and muscle cells contract as the sarcomeres contract.
The Sliding Filament Model of Contraction
When signaled by a motor neuron, a skeletal muscle fiber contracts as the thin filaments are pulled and then slide past the thick filaments within the fiber’s sarcomeres. This process is known as the sliding filament model of muscle contraction (Figure 6). The sliding can only occur when myosin-binding sites on the actin filaments are exposed by a series of steps that begins with Ca++ entry into the sarcoplasm.

Figure 6. The Sliding Filament Model of Muscle Contraction. When a sarcomere contracts, the Z lines move closer together, and the I band becomes smaller. The A band stays the same width. At full contraction, the thin and thick filaments overlap.
Tropomyosin is a protein that winds around the chains of the actin filament and covers the myosin-binding sites to prevent actin from binding to myosin. Tropomyosin binds to troponin to form a troponin-tropomyosin complex. The troponin-tropomyosin complex prevents the myosin “heads” from binding to the active sites on the actin microfilaments. Troponin also has a binding site for Ca++ ions.
To initiate muscle contraction, tropomyosin has to expose the myosin-binding site on an actin filament to allow cross-bridge formation between the actin and myosin microfilaments. The first step in the process of contraction is for Ca++ to bind to troponin so that tropomyosin can slide away from the binding sites on the actin strands. This allows the myosin heads to bind to these exposed binding sites and form cross-bridges. The thin filaments are then pulled by the myosin heads to slide past the thick filaments toward the center of the sarcomere. But each head can only pull a very short distance before it has reached its limit and must be “re-cocked” before it can pull again, a step that requires ATP.
ATP and Muscle Contraction
For thin filaments to continue to slide past thick filaments during muscle contraction, myosin heads must pull the actin at the binding sites, detach, re-cock, attach to more binding sites, pull, detach, re-cock, etc. This repeated movement is known as the cross-bridge cycle. This motion of the myosin heads is similar to the oars when an individual rows a boat: The paddle of the oars (the myosin heads) pull, are lifted from the water (detach), repositioned (re-cocked) and then immersed again to pull (Figure 7). Each cycle requires energy, and the action of the myosin heads in the sarcomeres repetitively pulling on the thin filaments also requires energy, which is provided by ATP.

Figure 7. Skeletal Muscle Contraction. (a) The active site on actin is exposed as calcium binds to troponin. (b) The myosin head is attracted to actin, and myosin binds actin at its actin-binding site, forming the cross-bridge. (c) During the power stroke, the phosphate generated in the previous contraction cycle is released. This results in the myosin head pivoting toward the center of the sarcomere, after which the attached ADP and phosphate group are released. (d) A new molecule of ATP attaches to the myosin head, causing the cross-bridge to detach. (e) The myosin head hydrolyzes ATP to ADP and phosphate, which returns the myosin to the cocked position.
Cross-bridge formation occurs when the myosin head attaches to the actin while adenosine diphosphate (ADP) and inorganic phosphate (Pi) are still bound to myosin . Pi is then released, causing myosin to form a stronger attachment to the actin, after which the myosin head moves toward the M-line, pulling the actin along with it. As actin is pulled, the filaments move approximately 10 nm toward the M-line. This movement is called the power stroke, as movement of the thin filament occurs at this step . In the absence of ATP, the myosin head will not detach from actin.
One part of the myosin head attaches to the binding site on the actin, but the head has another binding site for ATP. ATP binding causes the myosin head to detach from the actin . After this occurs, ATP is converted to ADP and Pi by the intrinsic ATPase activity of myosin. The energy released during ATP hydrolysis changes the angle of the myosin head into a cocked position . The myosin head is now in position for further movement.
When the myosin head is cocked, myosin is in a high-energy configuration. This energy is expended as the myosin head moves through the power stroke, and at the end of the power stroke, the myosin head is in a low-energy position. After the power stroke, ADP is released; however, the formed cross-bridge is still in place, and actin and myosin are bound together. As long as ATP is available, it readily attaches to myosin, the cross-bridge cycle can recur, and muscle contraction can continue.
Note that each thick filament of roughly 300 myosin molecules has multiple myosin heads, and many cross-bridges form and break continuously during muscle contraction. Multiply this by all of the sarcomeres in one myofibril, all the myofibrils in one muscle fiber, and all of the muscle fibers in one skeletal muscle, and you can understand why so much energy (ATP) is needed to keep skeletal muscles working. In fact, it is the loss of ATP that results in the rigor mortis observed soon after someone dies. With no further ATP production possible, there is no ATP available for myosin heads to detach from the actin-binding sites, so the cross-bridges stay in place, causing the rigidity in the skeletal muscles.
Sources of ATP
ATP supplies the energy for muscle contraction to take place. In addition to its direct role in the cross-bridge cycle, ATP also provides the energy for the active-transport Ca++ pumps in the SR. Muscle contraction does not occur without sufficient amounts of ATP. The amount of ATP stored in muscle is very low, only sufficient to power a few seconds worth of contractions. As it is broken down, ATP must therefore be regenerated and replaced quickly to allow for sustained contraction. There are three mechanisms by which ATP can be regenerated: creatine phosphate metabolism, anaerobic glycolysis, fermentation and aerobic respiration.
Creatine phosphate is a molecule that can store energy in its phosphate bonds. In a resting muscle, excess ATP transfers its energy to creatine, producing ADP and creatine phosphate. This acts as an energy reserve that can be used to quickly create more ATP. When the muscle starts to contract and needs energy, creatine phosphate transfers its phosphate back to ADP to form ATP and creatine. This reaction is catalyzed by the enzyme creatine kinase and occurs very quickly; thus, creatine phosphate-derived ATP powers the first few seconds of muscle contraction. However, creatine phosphate can only provide approximately 15 seconds worth of energy, at which point another energy source has to be used (Figure 8).

Figure 8. Muscle Metabolism. Some ATP is stored in a resting muscle. As contraction starts, it is used up in seconds. More ATP is generated from creatine phosphate for about 15 seconds.
As the ATP produced by creatine phosphate is depleted, muscles turn to glycolysis as an ATP source. Glycolysis is an anaerobic (non-oxygen-dependent) process that breaks down glucose (sugar) to produce ATP; however, glycolysis cannot generate ATP as quickly as creatine phosphate. Thus, the switch to glycolysis results in a slower rate of ATP availability to the muscle. The sugar used in glycolysis can be provided by blood glucose or by metabolizing glycogen that is stored in the muscle. The breakdown of one glucose molecule produces two ATP and two molecules of pyruvic acid, which can be used in aerobic respiration or when oxygen levels are low, converted to lactic acid (Figure 9).

Figure 9. Glycolysis and Aerobic Respiration. Each glucose molecule produces two ATP and two molecules of pyruvic acid, which can be used in aerobic respiration or converted to lactic acid. If oxygen is not available, pyruvic acid is converted to lactic acid, which may contribute to muscle fatigue. This occurs during strenuous exercise when high amounts of energy are needed but oxygen cannot be sufficiently delivered to muscle.
If oxygen is available, pyruvic acid is used in aerobic respiration. However, if oxygen is not available, pyruvic acid is converted to lactic acid, which may contribute to muscle fatigue. This conversion allows the recycling of the enzyme NAD+ from NADH, which is needed for glycolysis to continue. This occurs during strenuous exercise when high amounts of energy are needed but oxygen cannot be sufficiently delivered to muscle. Glycolysis itself cannot be sustained for very long (approximately 1 minute of muscle activity), but it is useful in facilitating short bursts of high-intensity output. This is because glycolysis does not utilize glucose very efficiently, producing a net gain of two ATPs per molecule of glucose, and the end product of lactic acid, which may contribute to muscle fatigue as it accumulates.
Aerobic respiration is the breakdown of glucose or other nutrients in the presence of oxygen (O2) to produce carbon dioxide, water, and ATP. Approximately 95 percent of the ATP required for resting or moderately active muscles is provided by aerobic respiration, which takes place in mitochondria. The inputs for aerobic respiration include glucose circulating in the bloodstream, pyruvic acid, and fatty acids. Aerobic respiration is much more efficient than anaerobic glycolysis, producing approximately 36 ATPs per molecule of glucose versus four from glycolysis. However, aerobic respiration cannot be sustained without a steady supply of O2 to the skeletal muscle and is much slower (Figure 12). To compensate, muscles store small amount of excess oxygen in proteins call myoglobin, allowing for more efficient muscle contractions and less fatigue. Aerobic training also increases the efficiency of the circulatory system so that O2 can be supplied to the muscles for longer periods of time.

Figure 10. Cellular Respiration. Aerobic respiration is the breakdown of glucose in the presence of oxygen (O2) to produce carbon dioxide, water, and ATP. Approximately 95 percent of the ATP required for resting or moderately active muscles is provided by aerobic respiration, which takes place in mitochondria.
Muscle fatigue occurs when a muscle can no longer contract in response to signals from the nervous system. The exact causes of muscle fatigue are not fully known, although certain factors have been correlated with the decreased muscle contraction that occurs during fatigue. ATP is needed for normal muscle contraction, and as ATP reserves are reduced, muscle function may decline. This may be more of a factor in brief, intense muscle output rather than sustained, lower intensity efforts. Lactic acid buildup may lower intracellular pH, affecting enzyme and protein activity. Imbalances in Na+ and K+ levels as a result of membrane depolarization may disrupt Ca++ flow out of the SR. Long periods of sustained exercise may damage the SR and the sarcolemma, resulting in impaired Ca++ regulation.
Intense muscle activity results in an oxygen debt, which is the amount of oxygen needed to compensate for ATP produced without oxygen during muscle contraction. Oxygen is required to restore ATP and creatine phosphate levels, convert lactic acid to pyruvic acid, and, in the liver, to convert lactic acid into glucose or glycogen. Other systems used during exercise also require oxygen, and all of these combined processes result in the increased breathing rate that occurs after exercise. Until the oxygen debt has been met, oxygen intake is elevated, even after exercise has stopped.
Relaxation of a Skeletal Muscle
Relaxing skeletal muscle fibers, and ultimately, the skeletal muscle, begins with the motor neuron, which stops releasing its chemical signal, ACh, into the synapse at the NMJ. The muscle fiber will repolarize, which closes the gates in the SR where Ca++ was being released. ATP-driven pumps will move Ca++ out of the sarcoplasm back into the SR. This results in the “reshielding” of the actin-binding sites on the thin filaments. Without the ability to form cross-bridges between the thin and thick filaments, the muscle fiber loses its tension and relaxes.
Candela Citations
- Biology 2e. Provided by: OpenStax. Located at: https://openstax.org/details/books/biology-2e. License: CC BY: Attribution. License Terms: Download for free at http://cnx.org/contents/8d50a0af-948b-4204-a71d-4826cba765b8@8.19
