Learning Objectives
- Determine the molecular mass of a molecule.
- Predict the general shape of a simple covalent molecule.
Unlike the ions in ionic compounds, which are arranged in lattices called crystals, molecules of covalent compounds exist as discrete units with a characteristic mass and a certain three-dimensional shape.
Molecular Mass
The molecular mass of a molecule is simply the sum of the masses of the atoms in its formula. As with formula masses, it is important that you keep track of the number of atoms of each element in the molecular formula to obtain the correct molecular mass. (For more information about formula masses, see Chapter 3 “Ionic Bonding and Simple Ionic Compounds”, Section 3.5 “Formula Mass”.)
Example 7
What is the molecular mass of each covalent compound?
- H2O
- C6H6
- NO2
- N2O5
Solution
Skill-Building Exercise
What is the molecular mass of each covalent compound?
- C2H2
- CO
- CO2
- BF3
Molecular Shape: VSEPR Theory
Unlike ionic compounds, with their extended crystal lattices, covalent molecules are discrete units with specific three-dimensional shapes. The shape of a molecule is determined by the fact that covalent bonds, which are composed of negatively charged electrons, and any lone pairs of electrons tend to repel one another. This concept is called the valence shell electron pair repulsion (VSEPR) theory. For example, the two covalent bonds in BeCl2 stay as far from each other as possible, ending up 180° apart from each other. The result is a linear molecule:

The three covalent bonds in BF3 repel each other to form 120° angles in a plane, in a shape called trigonal planar:

The molecules BeCl2 and BF3 actually violate the octet rule; however, such exceptions will not be discussed in this text.
Note
Try sticking three toothpicks into a marshmallow or a gumdrop and see if you can find different positions where your “bonds” are farther apart than the planar 120° orientation.
The four covalent bonds in CCl4 arrange themselves three dimensionally, pointing toward the four corners of a tetrahedron and making bond angles of 109.5°:

The shape of a molecule with lone electron pair(s) around the central atom is influenced by both the bonds and the lone pairs. For example, NH3 has one lone electron pair and three bonded electron pairs. These four electron pairs repel each other and adopt a tetrahedral electron arrangement:
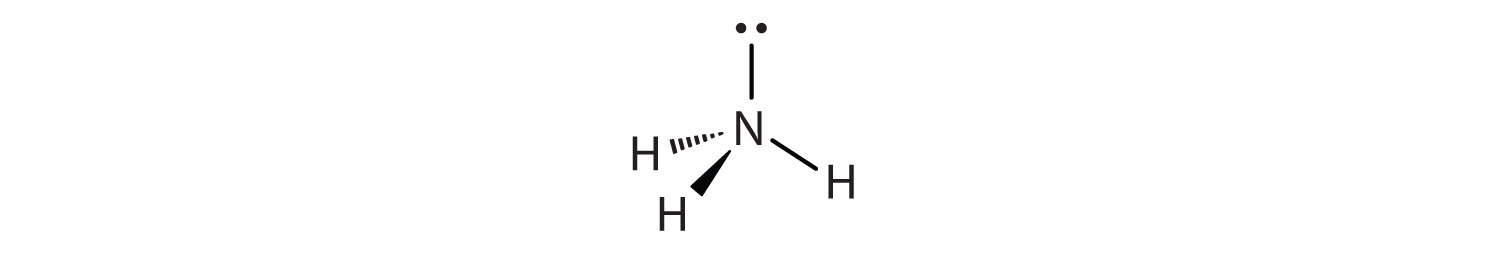
However, the shape of the molecule is described in terms of the positions of the atoms, not the lone electron pairs. Thus, NH3 is said to have a trigonal pyramidal molecular shape.
Similarly, H2O has two lone pairs of electrons around the central oxygen atom, in addition to the two bonded pairs:

Although the four electron pairs adopt a tetrahedral electron arrangement due to repulsion, the shape of the molecule is described by the positions of the atoms only. The shape of an H2O molecule is bent.
In determining the shapes of molecules, it is useful to first determine the Lewis diagram for a molecule. The shapes of molecules with multiple bonds are determined by treating the multiple bonds as one bond. Thus, CH2O has a shape similar to that of BF3. With three bonding groups around the central atom and no lone pairs, the CH2O molecule has a trigonal planar shape.

Example 8
Describe the shape of each molecule.
- PCl3
- CO2
Solution
Skill-Building Exercise
Describe the shape of each molecule.
-
CBr4
-
BCl3
Concept Review Exercises
-
How do you determine the molecular mass of a covalent compound?
-
How do you determine the shape of a molecule?
Key Takeaways
- A molecule has a certain mass, called the molecular mass.
- Simple molecules have geometries that can be determined from VSEPR theory.
Exercises
-
What is the molecular mass of each compound?
- H2S
- N2O4
- ICl3
- HCl
-
What is the molecular mass of each compound?
- O2F2
- CCl4
- C6H6
- SO3
-
Aspirin (C9H8O4) is a covalent compound. What is its molecular mass?
-
Cholesterol (C27H46O) is a biologically important compound. What is its molecular mass?
-
What is the shape of each molecule?
- H2S
- COCl2
- SO2
-
What is the shape of each molecule?
- NBr3
- SF2
- SiH4
Candela Citations
- The Basics of General, Organic, and Biological Chemistry v. 1.0. Provided by: Saylor Academy. Located at: https://saylordotorg.github.io/text_the-basics-of-general-organic-and-biological-chemistry/. License: CC BY-NC: Attribution-NonCommercial. License Terms: This text was adapted by Saylor Academy under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License without attribution as requested by the work's original creator or licensor.


