Esters have the general formula RCOOR′, where R may be a hydrogen atom, an alkyl group, or an aryl group, and R′ may be an alkyl group or an aryl group but not a hydrogen atom. (If it were hydrogen atom, the compound would be a carboxylic acid.) Figure 15.4 “The Structure of Esters” shows models for two common esters.

Figure 15.4 The Structure of Esters. Esters feature a carbon-to-oxygen double bond in which the carbon is also singly bonded to a second oxygen atom, which is then joined to an alkyl or an aryl group. The esters shown here are ethyl acetate (a) and methyl butyrate (b). (Note color scheme: Green = H, blue = C, red = O)
Esters occur widely in nature. Unlike carboxylic acids, esters generally have pleasant odors and are often responsible for the characteristic fragrances of fruits and flowers. Once a flower or fruit has been chemically analyzed, flavor chemists can attempt to duplicate the natural odor or taste. Both natural and synthetic esters are used in perfumes and as flavoring agents.
Note
Fats and vegetable oils are esters of long-chain fatty acids and glycerol. Esters of phosphoric acid are of the utmost importance to life. (For more information about fats/oils and esters, see Chapter 17 “Lipids”, Section 17.2 “Fats and Oils”, and Section 15.10 “Esters of Phosphoric Acid”, respectively.)
Names of Esters
Although esters are covalent compounds and salts are ionic, esters are named in a manner similar to that used for naming salts. The group name of the alkyl or aryl portion is given first and is followed by the name of the acid portion. It may be helpful to draw the structural formula in order to determine the alkyl/aryl and acid portions. In both common and International Union of Pure and Applied Chemistry (IUPAC) nomenclature, the –ic ending of the parent acid is replaced by the suffix –ate (Table 15.3 “Nomenclature of Esters”).
| Table 15.3 Nomenclature of Esters | ||
|---|---|---|
| Condensed Structural Formula | Common Name | IUPAC Name |
| HCOOCH3 | methyl formate | methyl methanoate |
| CH3COOCH3 | methyl acetate | methyl ethanoate |
| CH3COOCH2CH3 | ethyl acetate | ethyl ethanoate |
| CH3CH2COOCH2CH3 | ethyl propionate | ethyl propanoate |
| CH3CH2CH2COOCH(CH3)2 | isopropyl butyrate | isopropyl butanoate |
 |
ethyl benzoate | ethyl benzoate |
Example 5
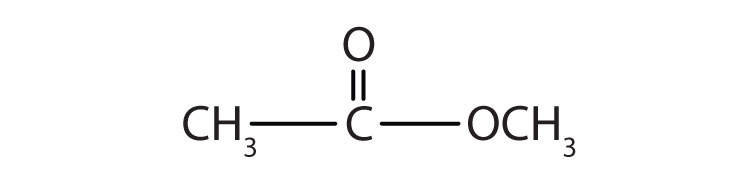
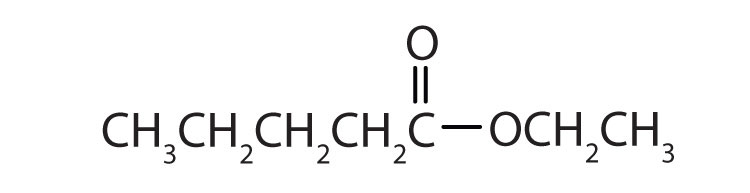
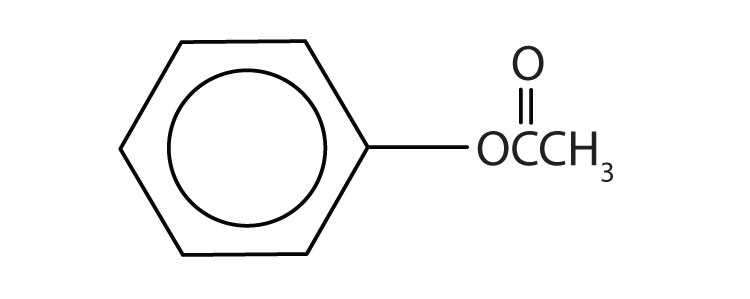
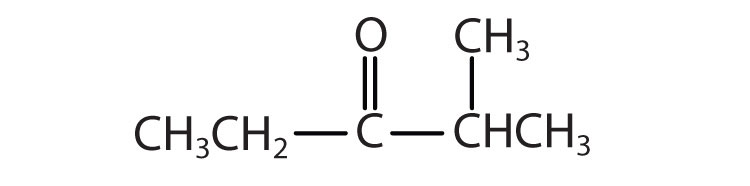
Give the common and IUPAC names for each compound.
Skill-Building Exercise
Give the common and IUPAC names for each compound.
Example 6
Draw the structure for ethyl pentanoate.
Skill-Building Exercise
-
Draw the structure for phenyl pentanoate.
Concept Review Exercises
-
From what carboxylic acid and what alcohol can isopropyl hexanoate be made?
-
From what carboxylic acid and what alcohol can cyclobutyl butyrate be made?
Key Takeaway
- An ester has an OR group attached to the carbon atom of a carbonyl group.
Exercises
-
Draw the structure for each compound.
- methyl acetate
- ethyl pentanoate
- phenyl acetate
- isopropyl propionate
-
Draw the structure for each compound.
- ethyl hexanoate
- ethyl benzoate
- phenyl benzoate
- ethyl 3-methylhexanoate
-
Name each compound with both the common name and the IUPAC name.
-
-
Name each compound with both the common name and the IUPAC name.
-
Candela Citations
- The Basics of General, Organic, and Biological Chemistry v. 1.0. Provided by: Saylor Academy. Located at: https://saylordotorg.github.io/text_the-basics-of-general-organic-and-biological-chemistry/. License: CC BY-NC: Attribution-NonCommercial. License Terms: This text was adapted by Saylor Academy under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License without attribution as requested by the work's original creator or licensor.