Learning Outcomes
- Describe how bones develop, grow, and repair
Ossification, or osteogenesis, is the process of bone formation by osteoblasts. Ossification is distinct from the process of calcification; whereas calcification takes place during the ossification of bones, it can also occur in other tissues. Ossification begins approximately six weeks after fertilization in an embryo. Before this time, the embryonic skeleton consists entirely of fibrous membranes and hyaline cartilage. The development of bone from fibrous membranes is called intramembranous ossification; development from hyaline cartilage is called endochondral ossification. Bone growth continues until approximately age 25. Bones can grow in thickness throughout life, but after age 25, ossification functions primarily in bone remodeling and repair.
Intramembranous Ossification
Intramembranous ossification is the process of bone development from fibrous membranes. It is involved in the formation of the flat bones of the skull, the mandible, and the clavicles. Ossification begins as mesenchymal cells form a template of the future bone. They then differentiate into osteoblasts at the ossification center. Osteoblasts secrete the extracellular matrix and deposit calcium, which hardens the matrix. The non-mineralized portion of the bone or osteoid continues to form around blood vessels, forming spongy bone. Connective tissue in the matrix differentiates into red bone marrow in the fetus. The spongy bone is remodeled into a thin layer of compact bone on the surface of the spongy bone.
Endochondral Ossification
Endochondral ossification is the process of bone development from hyaline cartilage. All of the bones of the body, except for the flat bones of the skull, mandible, and clavicles, are formed through endochondral ossification.
In long bones, chondrocytes form a template of the hyaline cartilage diaphysis. Responding to complex developmental signals, the matrix begins to calcify. This calcification prevents diffusion of nutrients into the matrix, resulting in chondrocytes dying and the opening up of cavities in the diaphysis cartilage. Blood vessels invade the cavities, and osteoblasts and osteoclasts modify the calcified cartilage matrix into spongy bone. Osteoclasts then break down some of the spongy bone to create a marrow, or medullary, cavity in the center of the diaphysis. Dense, irregular connective tissue forms a sheath (periosteum) around the bones. The periosteum assists in attaching the bone to surrounding tissues, tendons, and ligaments. The bone continues to grow and elongate as the cartilage cells at the epiphyses divide.
In the last stage of prenatal bone development, the centers of the epiphyses begin to calcify. Secondary ossification centers form in the epiphyses as blood vessels and osteoblasts enter these areas and convert hyaline cartilage into spongy bone. Until adolescence, hyaline cartilage persists at the epiphyseal plate (growth plate), which is the region between the diaphysis and epiphysis that is responsible for the lengthwise growth of long bones (Figure 1).
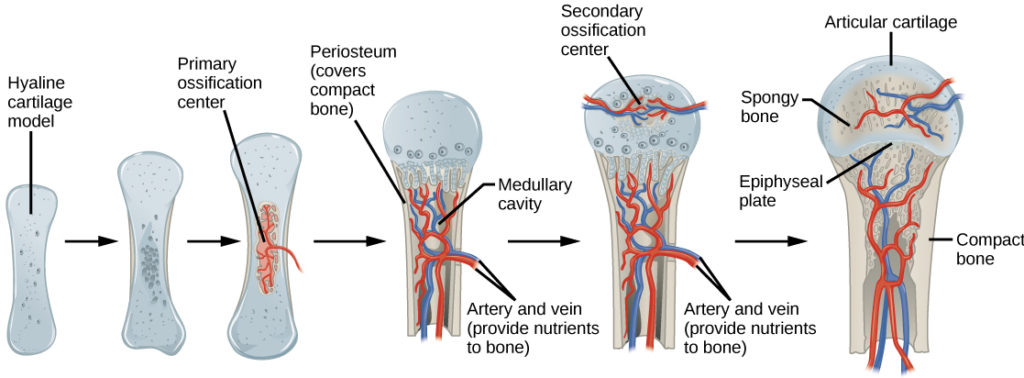
Figure 1. Endochondral ossification is the process of bone development from hyaline cartilage. The periosteum is the connective tissue on the outside of bone that acts as the interface between bone, blood vessels, tendons, and ligaments.
Growth of Bone
Long bones continue to lengthen, potentially until adolescence, through the addition of bone tissue at the epiphyseal plate. They also increase in width through appositional growth.
Lengthening of Long Bones
Chondrocytes on the epiphyseal side of the epiphyseal plate divide; one cell remains undifferentiated near the epiphysis, and one cell moves toward the diaphysis. The cells, which are pushed from the epiphysis, mature and are destroyed by calcification. This process replaces cartilage with bone on the diaphyseal side of the plate, resulting in a lengthening of the bone.
Long bones stop growing at around the age of 18 in females and the age of 21 in males in a process called epiphyseal plate closure. During this process, cartilage cells stop dividing and all of the cartilage is replaced by bone. The epiphyseal plate fades, leaving a structure called the epiphyseal line or epiphyseal remnant, and the epiphysis and diaphysis fuse.
Thickening of Long Bones
Appositional growth is the increase in the diameter of bones by the addition of bony tissue at the surface of bones. Osteoblasts at the bone surface secrete bone matrix, and osteoclasts on the inner surface break down bone. The osteoblasts differentiate into osteocytes. A balance between these two processes allows the bone to thicken without becoming too heavy.
Bone Remodeling and Repair
Bone renewal continues after birth into adulthood. Bone remodeling is the replacement of old bone tissue by new bone tissue. It involves the processes of bone deposition by osteoblasts and bone resorption by osteoclasts. Normal bone growth requires vitamins D, C, and A, plus minerals such as calcium, phosphorous, and magnesium. Hormones such as parathyroid hormone, growth hormone, and calcitonin are also required for proper bone growth and maintenance.
Bone turnover rates are quite high, with five to seven percent of bone mass being recycled every week. Differences in turnover rate exist in different areas of the skeleton and in different areas of a bone. For example, the bone in the head of the femur may be fully replaced every six months, whereas the bone along the shaft is altered much more slowly.

Figure 2. After this bone is set, a callus will knit the two ends together. (credit: Bill Rhodes)
Bone remodeling allows bones to adapt to stresses by becoming thicker and stronger when subjected to stress. Bones that are not subject to normal stress, for example when a limb is in a cast, will begin to lose mass. A fractured or broken bone undergoes repair through four stages:
- Blood vessels in the broken bone tear and hemorrhage, resulting in the formation of clotted blood, or a hematoma, at the site of the break. The severed blood vessels at the broken ends of the bone are sealed by the clotting process, and bone cells that are deprived of nutrients begin to die.
- Within days of the fracture, capillaries grow into the hematoma, and phagocytic cells begin to clear away the dead cells. Though fragments of the blood clot may remain, fibroblasts and osteoblasts enter the area and begin to reform bone. Fibroblasts produce collagen fibers that connect the broken bone ends, and osteoblasts start to form spongy bone. The repair tissue between the broken bone ends is called the fibrocartilaginous callus, as it is composed of both hyaline and fibrocartilage (Figure 2). Some bone spicules may also appear at this point.
- The fibrocartilaginous callus is converted into a bony callus of spongy bone. It takes about two months for the broken bone ends to be firmly joined together after the fracture. This is similar to the endochondral formation of bone, as cartilage becomes ossified; osteoblasts, osteoclasts, and bone matrix are present.
- The bony callus is then remodelled by osteoclasts and osteoblasts, with excess material on the exterior of the bone and within the medullary cavity being removed. Compact bone is added to create bone tissue that is similar to the original, unbroken bone. This remodeling can take many months, and the bone may remain uneven for years.
Decalcification of Bones
Question: What effect does the removal of calcium and collagen have on bone structure?
Background: Conduct a literature search on the role of calcium and collagen in maintaining bone structure. Conduct a literature search on diseases in which bone structure is compromised.
Hypothesis: Develop a hypothesis that states predictions of the flexibility, strength, and mass of bones that have had the calcium and collagen components removed. Develop a hypothesis regarding the attempt to add calcium back to decalcified bones.
Test the hypothesis: Test the prediction by removing calcium from chicken bones by placing them in a jar of vinegar for seven days. Test the hypothesis regarding adding calcium back to decalcified bone by placing the decalcified chicken bones into a jar of water with calcium supplements added. Test the prediction by denaturing the collagen from the bones by baking them at 250°C for three hours.
Analyze the data: Create a table showing the changes in bone flexibility, strength, and mass in the three different environments.
Report the results: Under which conditions was the bone most flexible? Under which conditions was the bone the strongest?
Draw a conclusion: Did the results support or refute the hypothesis? How do the results observed in this experiment correspond to diseases that destroy bone tissue?
IN SUMMARY: Bone Growth and Development
Ossification is the process of bone formation by osteoblasts. Intramembranous ossification is the process of bone development from fibrous membranes. Endochondral ossification is the process of bone development from hyaline cartilage. Long bones lengthen as chondrocytes divide and secrete hyaline cartilage. Osteoblasts replace cartilage with bone. Appositional growth is the increase in the diameter of bones by the addition of bone tissue at the surface of bones. Bone remodeling involves the processes of bone deposition by osteoblasts and bone resorption by osteoclasts. Bone repair occurs in four stages and can take several months.
Try It
Candela Citations
- Biology 2e. Provided by: OpenStax. Located at: http://cnx.org/contents/185cbf87-c72e-48f5-b51e-f14f21b5eabd@10.8. License: CC BY: Attribution. License Terms: Access for free at https://openstax.org/books/biology-2e/pages/1-introduction
