Conversion of alcohols to alkyl halides, tosylates and mesylates
Synthetic organic chemists, when they want to convert an alcohol into a better leaving group, have several methods to choose from. One common strategy is to convert the alcohol into an alkyl chloride or bromide, using thionyl chloride or phosphorus tribromide:


We won’t worry yet about the mechanism for the thionyl chloride reaction, but the PBr3 reaction is thought to involve two successive SN2-like steps:

Notice that these reactions result in inversion of stereochemistry in the resulting alkyl halide (as long as pyridine is used as solvent/base with the SOCl2).
With tertiary alcohols, an SN2 mechanism is disfavored because of the sterically hindered alkyl group, so tertiary alkyl halides are usually prepared using the hydrogen halide such as HCl or HBr. The mechanism involves activation of the hydroxyl group via an acid-base step, followed by heterolysis & coordination (i.e., an SN1 process overall):
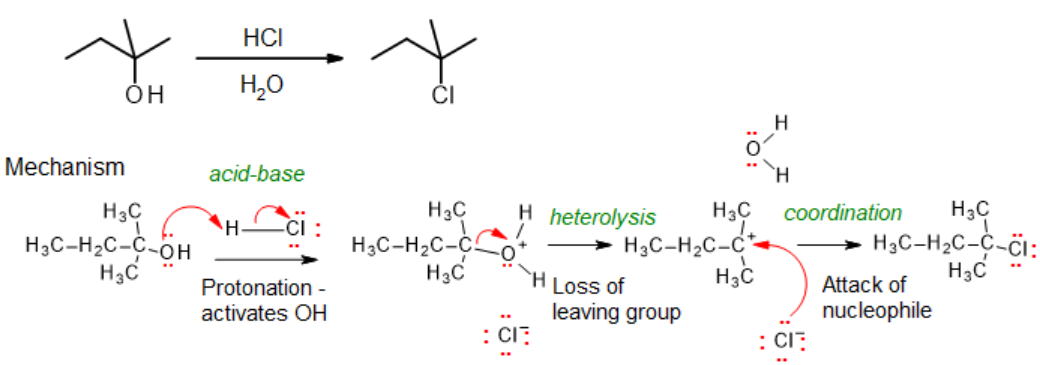
Reaction of 2-methylbutan-2-ol with HCl to produce 2-chloro-2-methylbutane.
An alternative type of leaving group is based on a sulfonate ester. We can easily transform an alcohol group into a sulfonate ester using para-toluenesulfonyl chloride (Ts-Cl) or methanesulfonyl chloride (Ms-Cl), creating what is termed an organic tosylate or mesylate:


Although it’s not shown in the scheme, this is normally done in the presence of a base such as triethylamine or pyridine, as we saw with the SOCl2 reaction. Notice, though, that unlike the halogenation reactions above, conversion of an alcohol to a tosylate or mesylate proceeds with retention of configuration at the electrophilic carbon.
Chlorides, bromides, and tosylate / mesylate groups are excellent leaving groups in nucleophilic substitution reactions, due to resonance delocalization of the developing negative charge on the leaving oxygen.

The laboratory synthesis of isopentenyl diphosphate – the ‘building block’ molecule used by nature for the construction of isoprenoid molecules such as cholesterol and b-carotene – was accomplished by first converting the alcohol into an organic tosylate (step 1), then displacing the tosylate group with an inorganic pyrophosphate nucleophile (step 2) (J. Org. Chem 1986, 51, 4768).

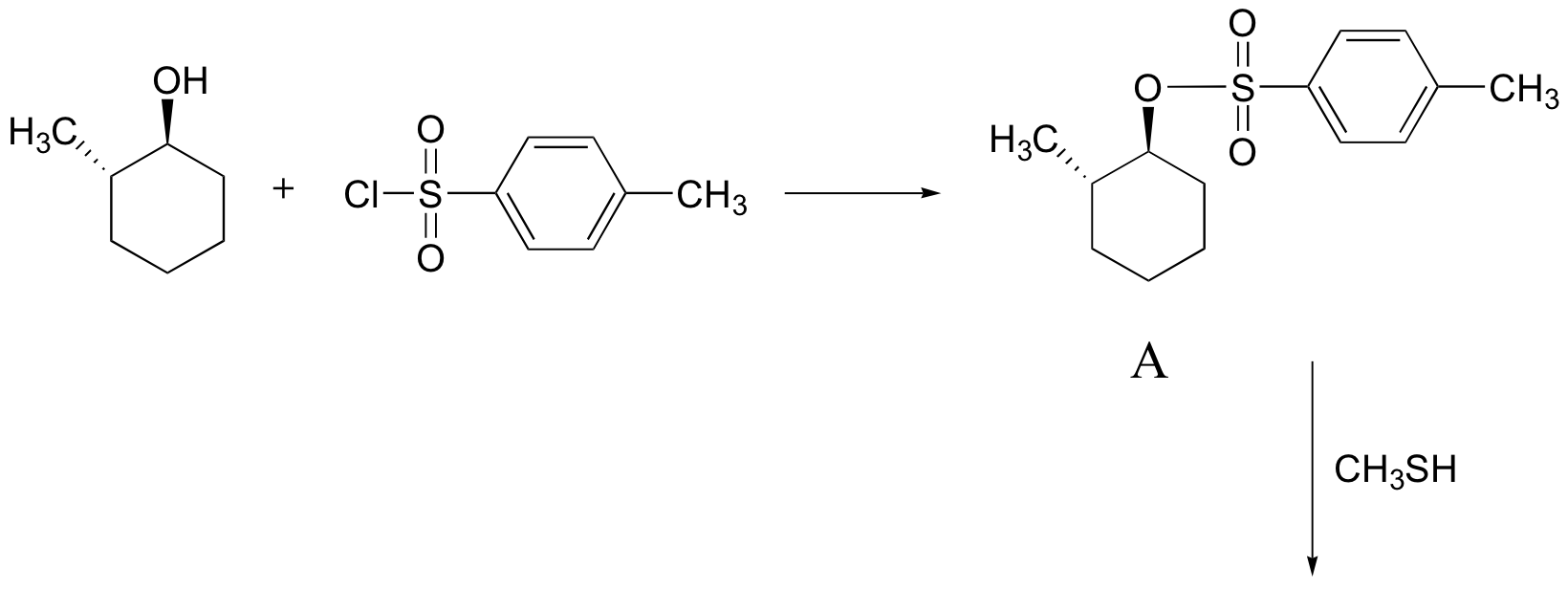
Exercise
Predict the structures of A and B in the following reaction:

Candela Citations
- 8.5: Leaving groups. Authored by: Tim Soderbergu00a0(University of Minnesota, Morris). Located at: https://chem.libretexts.org/Textbook_Maps/Organic_Chemistry/Book%3A_Organic_Chemistry_with_a_Biological_Emphasis_(Soderberg)/Chapter_08%3A_Nucleophilic_substitution_reactions_I/8.5%3A_Leaving_groups#8.5C:_Synthetic_parallel_-_conversion_of_alcohols_to_alkyl_halides.2C_tosylates_and_mesylates. Project: Chemistry LibreTexts. License: CC BY-NC-SA: Attribution-NonCommercial-ShareAlike