Learning Objectives
By the end of this section, you will be able to do the following:
- Explain pharmacogenomics
- Define polygenic
Introducing DNA sequencing and whole genome sequencing projects, particularly the Human Genome project, has expanded the applicability of DNA sequence information. Many fields, such as metagenomics, pharmacogenomics, and mitochondrial genomics are using genomics. Understanding and finding cures for diseases is the most common application of genomics.
Predicting Disease Risk at the Individual Level
Predicting disease risk involves screening currently healthy individuals by genome analysis at the individual level. Health care professionals can recommend intervention with lifestyle changes and drugs before disease onset. However, this approach is most applicable when the problem resides within a single gene defect. Such defects only account for approximately 5 percent of diseases in developed countries. Most of the common diseases, such as heart disease, are multi-factored or polygenic, which is a phenotypic characteristic that involves two or more genes, and also involve environmental factors such as diet. In April 2010, scientists at Stanford University published the genome analysis of a healthy individual (Stephen Quake, a scientist at Stanford University, who had his genome sequenced. The analysis predicted his propensity to acquire various diseases. The medical team performed a risk assessment to analyze Quake’s percentage of risk for 55 different medical conditions. The team found a rare genetic mutation, which showed him to be at risk for sudden heart attack. The results also predicted that Quake had a 23 percent risk of developing prostate cancer and a 1.4 percent risk of developing Alzheimer’s. The scientists used databases and several publications to analyze the genomic data. Even though genomic sequencing is becoming more affordable and analytical tools are becoming more reliable, researchers still must address ethical issues surrounding genomic analysis at a population level.
Art Connection

Figure 1. PCA3 is a gene that is expressed in prostate epithelial cells and overexpressed in cancerous cells. A high PCA3 concentration in urine is indicative of prostate cancer. The PCA3 test is a better indicator of cancer than the more well known PSA test, which measures the level of PSA (prostate-specific antigen) in the blood.
In 2011, the United States Preventative Services Task Force recommended against using the PSA test to screen healthy men for prostate cancer. Their recommendation is based on evidence that screening does not reduce the risk of death from prostate cancer. Prostate cancer often develops very slowly and does not cause problems, while the cancer treatment can have severe side effects. The PCA3 test is more accurate, but screening may still result in men who would not have been harmed by the cancer itself suffering side effects from treatment. What do you think? Should all healthy men receive prostate cancer screenings using the PCA3 or PSA test? Should people in general receive screenings to find out if they have a genetic risk for cancer or other diseases?
Pharmacogenomics and Toxicogenomics
Pharmacogenomics, or toxicogenomics, involves evaluating drug effectiveness and safety on the basis of information from an individual’s genomic sequence. We can study genomic responses to drugs using experimental animals (such as laboratory rats or mice) or live cells in the laboratory before embarking on studies with humans. Studying changes in gene expression could provide information about the transcription profile in the drug’s presence, which we can use as an early indicator of the potential for toxic effects. For example, genes involved in cellular growth and controlled cell death, when disturbed, could lead to cancerous cell growth. Genome-wide studies can also help to find new genes involved in drug toxicity. Medical professionals can use personal genome sequence information to prescribe medications that will be most effective and least toxic on the basis of the individual patient’s genotype. The gene signatures may not be completely accurate, but medical professionals can test them further before pathologic symptoms arise.
Microbial Genomics: Metagenomics
Traditionally, scholars have taught microbiology with the view that it is best to study microorganisms under pure culture conditions. This involves isolating a single cell type and culturing it in the laboratory. Because microorganisms can go through several generations in a matter of hours, their gene expression profiles adapt to the new laboratory environment very quickly. In addition, the vast majority of bacterial species resist culturing in isolation. Most microorganisms do not live as isolated entities, but in microbial communities or biofilms. For all of these reasons, pure culture is not always the best way to study microorganisms. Metagenomics is the study of the collective genomes of multiple species that grow and interact in an environmental niche. Metagenomics can be used to identify new species more rapidly and to analyze the effect of pollutants on the environment ((Figure)).
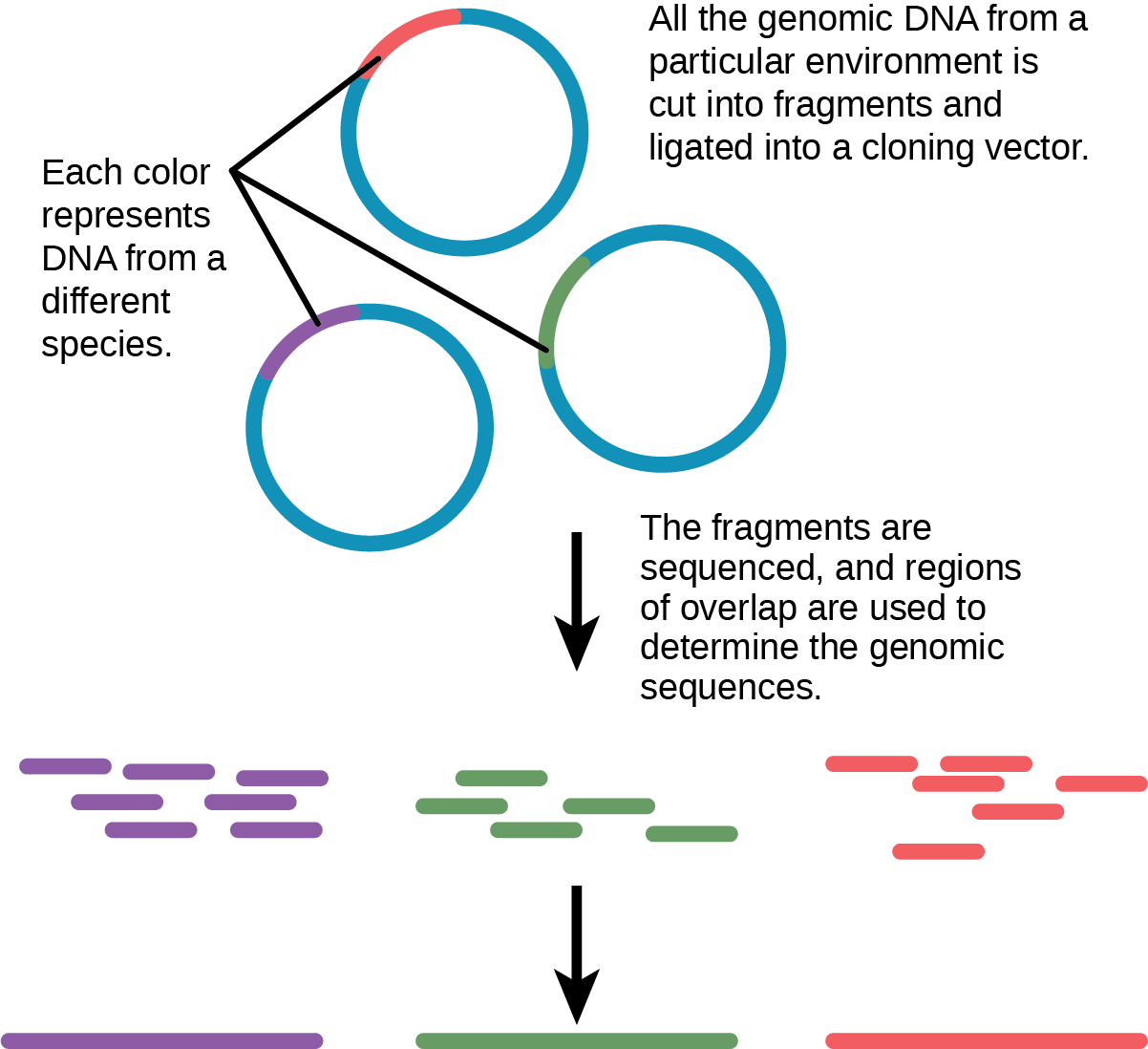
Figure 2. Metagenomics involves isolating DNA from multiple species within an environmental niche.
Microbial Genomics: Creation of New Biofuels
Knowledge of the genomics of microorganisms is being used to find better ways to harness biofuels from algae and cyanobacteria. The primary sources of fuel today are coal, oil, wood, and other plant products, such as ethanol. Although plants are renewable resources, there is still a need to find more alternative renewable sources of energy to meet our population’s energy demands. The microbial world is one of the largest resources for genes that encode new enzymes and produce new organic compounds, and it remains largely untapped. Microorganisms are used to create products, such as enzymes that are used in research, antibiotics, and other antimicrobial mechanisms. Microbial genomics is helping to develop diagnostic tools, improved vaccines, new disease treatments, and advanced environmental cleanup techniques.
Mitochondrial Genomics
Mitochondria are intracellular organelles that contain their own DNA. Mitochondrial DNA mutates at a rapid rate and scientists often use it to study evolutionary relationships. Another feature that makes studying the mitochondrial genome interesting is that the mitochondrial DNA in most multicellular organisms passes from the mother during the fertilization process. For this reason, scientists often use mitochondrial genomics to trace genealogy.
Experts have used information and clues from DNA samples at crime scenes as evidence in court cases, and they have used genetic markers in forensic analysis. Genomic analysis has also become useful in this field. The first publication showcasing the first use of genomics in forensics came out in 2001. It was a collaborative attempt between academic research institutions and the FBI to solve the mysterious cases of anthrax communicated via the US Postal Service. Using microbial genomics, researchers determined that the culprit used a specific anthrax strain in all the mailings.
Genomics in Agriculture
Genomics can reduce the trials and failures involved in scientific research to a certain extent, which could improve agricultural crop yield quality and quantity. Linking traits to genes or gene signatures helps improve crop breeding to generate hybrids with the most desirable qualities. Scientists use genomic data to identify desirable traits, and then transfer those traits to a different organism. Researchers are discovering how genomics can improve agricultural production’s quality and quantity. For example, scientists could use desirable traits to create a useful product or enhance an existing product, such as making a drought-sensitive crop more tolerant of the dry season.
Section Summary
Imagination is the only barrier to the applicability of genomics. Researchers are applying genomics to most fields of biology. They use it for personalized medicine, prediction of disease risks at an individual level, studying drug interactions before conducting clinical trials, and studying microorganisms in the environment as opposed to the laboratory. They are also applying it to developments such as generating new biofuels, genealogical assessment using mitochondria, advances in forensic science, and improvements in agriculture.
Art Connections
(Figure) In 2011, the United States Preventative Services Task Force recommended against using the PSA test to screen healthy men for prostate cancer. Their recommendation is based on evidence that screening does not reduce the risk of death from prostate cancer. Prostate cancer often develops very slowly and does not cause problems, while the cancer treatment can have severe side effects. The PCA3 test is considered to be more accurate, but screening may still result in men who would not have been harmed by the cancer itself suffering side effects from treatment. What do you think? Should all healthy men be screened for prostate cancer using the PCA3 or PSA test? Should people in general be screened to find out if they have a genetic risk for cancer or other diseases?
Review Questions
Genomics can be used in agriculture to:
- generate new hybrid strains
- improve disease resistance
- improve yield
- all of the above
Genomics can be used on a personal level to:
- decrease transplant rejection
- predict genetic diseases that a person may have inherited
- determine the risks of genetic diseases for an individual’s children
- all of the above
Free Response
Explain why metagenomics is probably the most revolutionary application of genomics.
How can genomics be used to predict disease risk and treatment options?
Glossary
- metagenomics
- study of multiple species’ collective genomes that grow and interact in an environmental niche
- pharmacogenomics
- study of drug interactions with the genome or proteome; also called toxicogenomics
- polygenic
- phenotypic characteristic caused by two or more genes
- pure culture
- growth of a single cell type in the laboratory
Candela Citations
- Biology 2e. Provided by: OpenStax. Located at: https://openstax.org/details/books/biology-2e. License: CC BY: Attribution. License Terms: Download for free at http://cnx.org/contents/8d50a0af-948b-4204-a71d-4826cba765b8@8.19
