Addition of strong acids to alkynes is quite similar to the addition of strong acids to alkenes. The regiochemistry follows Markovnikov’s Rule, but the stereochemistry is often different. Addition to alkenes is usually not stereospecific, whereas alkynes usually undergo anti addition.
Now we will take a look at electrophilic addition reactions, particularly of alkynes. The reaction mechanisms, as you will notice, are quite similar to the electrophilic addition reactions of alkenes. The triple bonds of alkynes, because of its high electron density, are easily attacked by electrophiles, but are less reactive than alkenes due to the compact C-C electron cloud.As with electrophilic addition to unsymmetrical alkenes, the Markovnikov rule is followed, adding the electrophile to the less substituted carbon.
For simplicity, we will show the mechanism going via a vinyl carbocation, by analogy with the alkyl carbocation formed when alkenes react. In fact, the vinyl carbocation is usually to unstable to be a real intermediate; the actual intermediate is more like a complex of the electrophile with the alkyne, which begins to place a partial positive charge on the nearby carbon.
Here we will go through the following reactions listed below to learn the mechanisms behind these electrophilic additions of alkynes: (1) HX Addition to alkenes, (2) Halogenation of alkynes and (3) Hydration of alkynes.
Figure

The addition of an electrophile to either an alkene or an alkyne will undergo the same steps listed below.
- Start with a reactant (either an alkene or an alkyne), which has π electrons. An electron pair moves from the π bond to the electrophilic proton to form a new covalent bond this is the electrophilic addition elementary step. A carbocation intermediate forms on the more stable carbon. As shown in the diagram above, the intermediate consists of hydrogen covalently bonded to the carbon and a positive charge on the other carbon.
- The addition of the halide ion to the positive charged intermediate forms the second covalent bond giving you the haloalkane product (as seen on the left of the above diagram) and a haloalkene product (as seen on the right of the above diagram). The halide ion acts as a nucleophile, which is attracted to the positive charged carbon because it has electrons to donate or share. So when the nucleophilic halide (represented as X–) attacks, it aims for the positively charged carbon resulting in that second covalent bond as seen above.
Make Note: The way to determine where the addition of the proton and the halide takes place is based on Markovnikov’s Rule, which states that the proton adds onto the carbon with the most hydrogens and the halogen prefers the most substituted carbon.
Now let’s take a look at our first reaction, the addition of Hydrogen Halide.
Reaction 1: Addition of hydrogen halide to an alkyne
Summary: Reactivity order of hydrogen halides: HI > HBr > HCl > HF.
Follows Markovnikov’s rule:
- Hydrogen adds to the carbon with the greatest number of hydrogens, the halogen adds to the carbon with fewer hydrogens.
- Protonation occurs on the more stable carbocation. With the addition of HX, haloalkenes form.
- With the addition of excess HX, you get anti addition forming a geminal dihaloalkane.
Addition of a HX to an internal alkyne
As described in Figure 1, the π electrons will attack the hydrogen of the HBr and because this is a symmetric molecule it will add to either carbon at a roughly equal rate. Once the hydrogen is covalently bonded to one of the carbons, you will get a carbocation intermediate (not shown, but will look the same as depicted in the figure above) on the other carbon.
The final step is the addition of the bromide, which is a good nucleophile because it has electrons to donate or share. Bromide, therefore, attacks the carbocation intermediate placing the Br on that carbon. As a result, you get 2-bromobutene from your 2-butyne reactant, as shown below.
Figure 2

Now, what if you have excess HBr?
Addition due to excess HX present yields a geminal dihaloalkane
Figure 3

Here, the electrophilic addition proceeds with the same steps used to achieve the product in addition of a HX to an internal alkyne. The π electrons attacked the hydrogen, adding it to the carbon on the left (shown in blue). The hydrogen is added to the left hand carbon forming a carbocation on the carbon with the Br, since this is stabilized by type III resonance. Then the carbocation intermediate is attacked by the bromide resulting in a dihaloalkane product.
Addition of HX to Terminal Alkyne
- Here is an addition of HBr to an asymmetric molecule.
- First, try to make sense of how the reactant went to product and then take a look at the mechanism.

The π electrons are attacking the hydrogen, depicted by the electron pushing arrows and the bromine gains a negative charge. The carbocation intermediate forms a positive charge on the left carbon after the hydrogen was added to the carbon with the most hydrogen substituents.

The bromine, which now exists as bromide with a negative charge, attacks the positively charged carbocation forming the final product with the nucleophile (Br) on the more substituted carbon.
Addition with excess HBr present

Reaction: Halogenation of Alkynes
Summary:

- Stereoselectivity: anti addition
- Reaction proceeds via cyclic halonium ion
Addition of Br2
- The addition of Br2 to an alkyne is analogous to adding Br2 to an alkene.
- Once Br2 approaches the nucleophilic alkyne, it becomes polarized.
- The π electrons, from the triple bond, can now attack the polarized bromine forming a C-Br bond and displacing the bromide ion.
- Now, you will get an intermediate bromonium ion, which will immediately react with the bromide ion giving you the dibromo product.

First, you see the polarized Br2 being attacked by the π electrons. Once you form the C-Br bond, the other bromine is released as a bromine ion. The intermediate here is a bromonium ion, which is electrophilic and reacts with the bromine ion giving you the dibromo product.
Reaction: Hydration of Alkynes
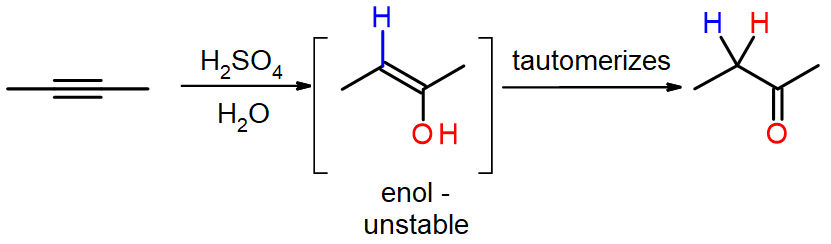
Summary: With the addition of water, alkynes can be hydrated to form enols that spontaneously tautomerize to ketones. Reaction is catalyzed by mercury ions. Follows Markovnikov’s Rule: Terminal alkynes give methyl ketones.
Figure 7

- The first step is an acid/base reaction where the π electrons of the triple bond act as a Lewis base and attack the proton, protonating the carbon that has more hydrogens. (Electrophilic addition)
- The second step is the attack of the nucleophilic water molecule on the electrophilic carbocation, which creates an oxonium ion. (Coordination)
- Next, the positively charged oxygen is deprotonated by water (which is acting as a base), generating an C=C bonded alcohol called an enol. (Acid-base)
- The enol then tautomerizes into a ketone, via a mechanism that will be covered in part 2 of this textbook. Tautomerization is a simultaneous proton and double bond shift, which goes from the enol form to the keto isomer form as shown above in Figure 7.
Now let’s look at some examples of hydration Reactions.
Hydration of terminal alkyne produces methyl ketones

Just as described in Figure 7 the π electrons will attack a proton, forming a carbocation, which then gets attacked by the nucleophilic water molecules. After deprotonation, we generate an enol, which then tautomerizes into the ketone form shown.
Hydration of but-1-yne
The π electrons of the triple bond attack the proton, which forms a covalent bond on the carbon with the most hydrogen substituents. This forms a carbocation, which is then attacked by the water molecule via a coordination step. This produces a positive charge on the oxygen, so another molecule of water is needed as a base to deprotonate the molecule and give a neutral product. The resultant enol is unstable, and it rapidly tautomerizes to the ketone (butanone).
Figure 9
Outside links

References
- Vollhardt. Schore, Organic Chemistry Structure and Function Fifth Edition, New York: W.H. Freeman and Company, 2007.
- Solomons, T. W. Graham, and Craig B. Fryhle. Organic Chemistry Eighth Edition. Maryland: John Wiley and Sons Incorporated, 2008.
Practice Problems
1) Reaction: Addition of hydrogen halide to an alkyne
1a) What product would result from the reaction of 1-pentyne with hydrogen bromide?

1b) Take your answer from 1a and add an excess of hydrogen bromide. What would be the product?

1c) What is the mechanism for this reaction?
2) Reaction: Addition of X2 to alkynes
2a) What product would result from the reaction of 1-pentyne with Br2?
.bmp?revision=1&size=bestfit&width=382&height=74#fixme#fixme)
2b) Take your answer from 2a and add an excess of Br2. What would be the product?

2c) What is the mechanism for this reaction?
3) Reaction: Hydration of alkynes
3a) If 1-Pentyne were to react with mercury(II) sulfate, water, and sulfuric acid, what would be the product?

4) What is the product when 3-methylbutyne reacts with HCl? Include in your answer:
a) the reaction mechanism
b) a clear explanation for why the hydrogen and chlorine bind to where they do
5) a) Draw the enol form of the ketone below.

b) Draw the keto form of the enol below.

Further reading
MasterOrganicChemistry


Carey 5th Ed Online

Videos


Candela Citations
- 11.1.3 Electrophilic Addition to Alkynes. Authored by: Prof. Hilton Weiss (Bard College), Aleksandra Milman. Located at: https://chem.libretexts.org/LibreTexts/Purdue/Purdue%3A_Chem_26605%3A_Organic_Chemistry_II_(Lipton)/Chapter_11.__Addition_to_pi_Systems/11.1%3A_Electrophilic_Addition/11.1.3_Electrophilic_Addition_to_Alkynes. Project: Chemistry LibreTexts. License: CC BY-NC-SA: Attribution-NonCommercial-ShareAlike